Phosphorus is relevant in the aquarium in only three forms: dissolved inorganic phosphate (DIP), solid inorganic precipitates (PIP) and organically bound in dissolved (DOP) or solid (POP) substances.
Various forms of life take up phosphorus in different forms. Inorganic phosphate can only be taken up by autotrophic organisms (plants, algae, zooxanthellae and some bacteria). Some specialised bacteria can also utilise solid phosphate salts (e.g. hydroxyapatite) in the soil interstitial space and in porous superstructures. These so-called phosphate pools can cause great difficulties. All other heterotrophic organisms (animals, many bacteria, fungi) can only utilise phosphorus organically bound (dissolved = DOP or particulate = POP).
Excessive phosphate concentrations affect the calcification of corals, especially in combination with high carbonate hardness. If there are increased phosphate concentrations, the carbonate hardness should not exceed 7.5°dKH. The phosphate concentrations given below are only to be understood as recommendations. Species-specific differences exist - very high phosphate concentrations above 1 mg/l can be tolerated by some stony corals. The most important rule in the matter of phosphate is: if concentration changes are made, then only very slowly over several weeks. When buying stony coral offshoots, especially SPS, make sure that the phosphate concentrations in the sales tank are similar to those in your own aquarium.
There are a number of ways to reduce or increase the phosphate concentration in the aquarium: see Links: Phosphate check list.
SPS aquariums: 0.05...0.2 mg/l phosphate
Reef aquariums, soft corals: 0.05...2 mg/l phosphate
Fish aquariums: 0.1...2 mg/l phosphate
Many animals can adapt to higher phosphate concentrations. However, severe changes are not well tolerated and should be prevented at all costs


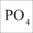

 Phosphate-X-globuli for phosphate removal
Phosphate-X-globuli for phosphate removal Phosphate adsorber with good cost-performance ratio
Phosphate adsorber with good cost-performance ratio