|
We all know osmotic processes. If you watch ripe black cherries it is possible that they will get ruptured or scared over places after a rain shower. The reason for it is the fact, that the skin of a cherry is a semipermeable membrane. Inside there is a solution with a high ionic concentration (cherry juice contains a lot of sugar and salts) and outside are rain drops with nearly no ions. The only way to balance the inside and outside concentrations is that water is flowing into the cherry - sugar and salts are too big to get through the cherry skin. But the cherry cannot grow so fast and therefore the inside migrating water raises the inside pressure - so called osmotic pressure - until the skin will rupture.
With reverse osmosis techique the principle of osmosis is inverted. At the high concentration side (feed water, e.g. tap water) is a pressure (pump or tap water pressure) that forces the water molecules against their natural migration through the membrane onto the pure water side (permeate side). The salts (e.g. hardnes, nitrate, silicic acid, residues of pesticides and medicine) and other substances (except gases) are nearly not able to pass the membrane because they are too big. So on the permeate side is nearly only water.
With the feed water a constant flow of new substances will reach the membrane. If you do not drain them they will accumulate and heavy soluble salts will precipitate. So a reverse osmosis unit will produce pure water (permeate) AND waste water (concentrate).
|



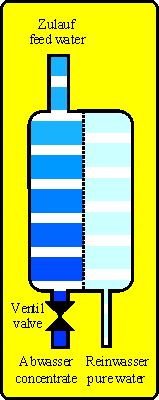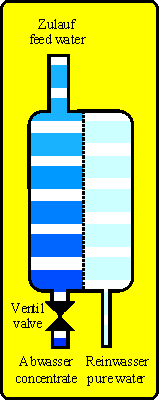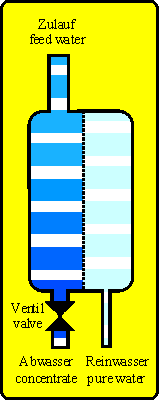

 Technical units driven with water may leak as a basic principle and can cause water damages. If secondary damages are possible the unit should be protected by using leaking detectors, drip pan or others.
Technical units driven with water may leak as a basic principle and can cause water damages. If secondary damages are possible the unit should be protected by using leaking detectors, drip pan or others. BasiTech Reverse Osmosis Control for small RO
BasiTech Reverse Osmosis Control for small RO TDS stick for testing the salt concentration
TDS stick for testing the salt concentration UV lamp for disinfecting fresh and RO water
UV lamp for disinfecting fresh and RO water