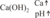-
Filter
- Chalk
- Skimmer ACF
- LSS tank "Basic"
- MultiFunctionFilter MF2
- Globuli Filter
- Pellet Filter POC
- Nitrate Filter ADN
- Neutralisierungsfilter NF
- Phosphate Filter PO4
- Fluidized Bed Filter FBF
- Activated Carbon Filter AK
- Zeolite Filter
- Trickling Filter TKF
- Sponge Loop Filter
- Ozone Reactor OZR
- CO2 reactor COR
- Gas filters
- Small Filter
- Empty Filter
- Filter Materials
- Filter Accessories
- UV lamps
- Additives
- Measuring
- Keeping/Breeding
- Water
- Pumps
- Fittings
- Installation
- Info
AquaCare-shop.de Info Encyclopaedia Seawater Filter Technique 51 Kalkwasser reactor
The lime water method of dripping dissolved calcium hydroxide into the aquarium water has several advantages:
- The calcium concentration is increased. If calcium levels are elevated, the method should be suspended.
- The pH value is raised slightly. Therefore, it is favorable to let the lime water drip in overnight.
- Phosphate reduction by precipitation to hydroxyapatite (poorly soluble phosphate salt). The method should not be used in case of phosphate deficiency.
- Increase of carbonate hardness (alkalinity): whether and how much carbonate hardness is produced depends on the free CO2 concentration in the water. In general, no carbonate hardness is produced.
If the lime water method is used, it is important that the freshly prepared calcium hydroxide solution does not come into contact with air, because the containing carbon dioxide makes the solution unusable. If lime water is used manually, it should be done with a closed canister - only a tiny vent is required. In the case of a lime water reactor, the entire system should be airtight to ensure prolonged storage. When comparing the different lime water reactor models, the maximum loading is the decisive criterion - the larger the possible loading, the longer the maintenance interval.
FAQ "Kalkwasserreaktor" (KWR - Calcium hydroxid): https://www.aquacare.de/index.php/en/aquaristic/aquaristic-info/faq.html#kwr
Dosing lime water "Kalkwasser": https://www.aquacare.de/index.php/en/aquaristic/aquaristic-info/hints-tricks/50-water-chemistry-physics/268-methods-lime-water.html
Total hardness / Alkalinity / Magnesium / KH / Acid capacity - an overview: https://www.aquacare.de/index.php/en/aquaristic/aquaristic-info/hints-tricks/50-water-chemistry-physics/250-hardness-overview.html
Methods for rising soluble calcium and hydrogen carbonate (alkalinity) in sea water: https://www.aquacare.de/index.php/en/aquaristic/aquaristic-info/hints-tricks/50-water-chemistry-physics/254-methods-hardness.html
Sodium Hydrogen Carbonate in the KWR: https://www.aquacare.de/index.php/en/aquaristic/aquaristic-info/hints-tricks/50-water-chemistry-physics/269-sodium-hydrogen-carbonate-in-kwr.html